Many drug actives are intensely bitter or have other aversive taste, aroma, texture or mouthfeel attributes. Unfortunately, these attributes are frequently unknown or inadequately addressed during development, resulting in drug products that may not be well tolerated by patients.
When drug products are not properly taste-masked, dosing compliance and health outcomes suffer. This is especially true for pediatric formulations or drugs for adults who have difficulty swallowing.
At Senopsys, we never forget that patients are people!
At Senopsys, we help reduce technical and market risks by doing what we do best – develop palatable drug products!
Senopsys scientists are experts in taste assessment, taste modulation, taste masking, excipient functionality and flavor construction.
We have decades of experience developing palatable liquid, powder and solid dosage forms of investigational and approved drugs.
Senopsys partners with pharmaceutical, biotechnology and drug delivery companies to develop palatable drug formulations that meet the needs of diverse patient populations
We are independent, objective and “technology agnostic” and do not license technology, sell ingredients or manufacture products. We are collaborative and routinely work with our client’s CDOs, CDMOs and CROs.
But Senopsys does more than just develop palatable drug products. We also provide our clients with early stage guidance on age-appropriate dosage forms, PIPs/PSPs, Target Product Profiles, taste masking technology and formulation approaches, data requirements, and more.
Senopsys is the only company that offers both a robust taste assessment analytics platform and a proven taste masking formulation development process.
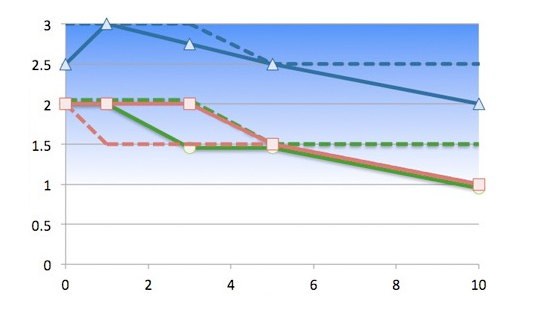
A client’s drug achieved strong positive clinical results in adults and a pediatric dosage form was urgently required. Senopsys’ initial taste assessment indicated that the API represented a difficult, multidimensional taste challenge, specifically strong intensity bitterness, tongue/throat burn and tannin mouthfeels that lingered for over 30 minutes…

How good is your knowledge of taste masking? Learn the truths behind the science and what it means for your development program.
Senopsys is the taste-masking development partner of choice for 15 of the top 25 global pharma companies as well as dozens of emerging and mid-size pharma and biotech companies.
Ask us questions, inquire about our services or schedule a “lunch and learn” seminar about the art and science of taste masking.